Get a Higher-Sensitivity Sample in a Few Simple Steps
We designed the MicroWash device to provide a simpler, safer patient testing experience with higher-sensitivity samples than traditional nasal swabs or saline irrigation methods. Our priority is ensuring unmatched comfort for users while giving labs higher-sensitivity testing results with fewer extraction errors.
Overview for Use
Our patented groundbreaking device revolutionizes respiratory infection nasal lavage sampling. Medical professionals get quicker, higher-sensitivity samples in multiple settings with increased throughput. Sampling is easy, safe, comfortable and consistent. Refer to the Complete IFU for detailed instructions.
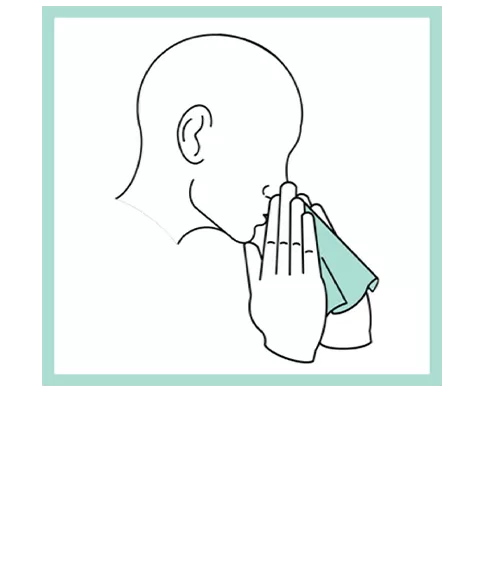


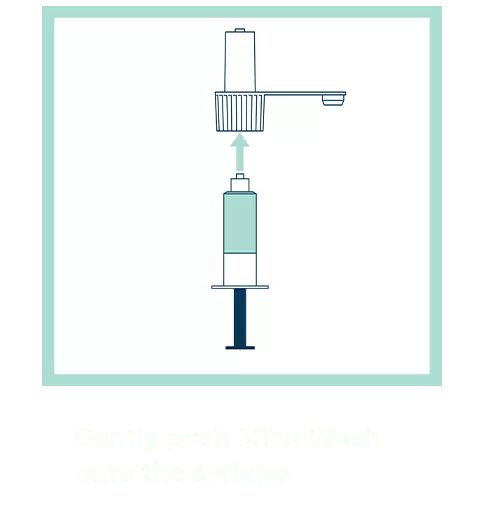
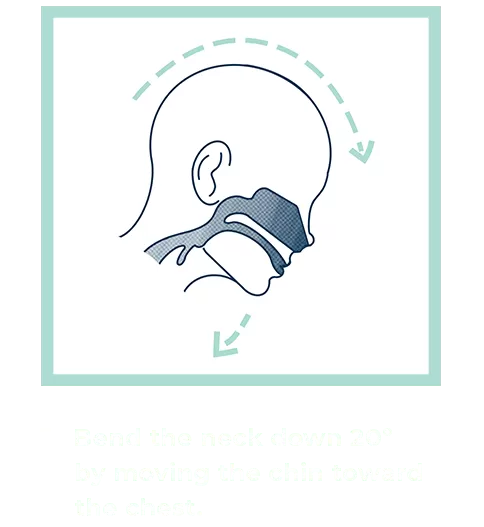



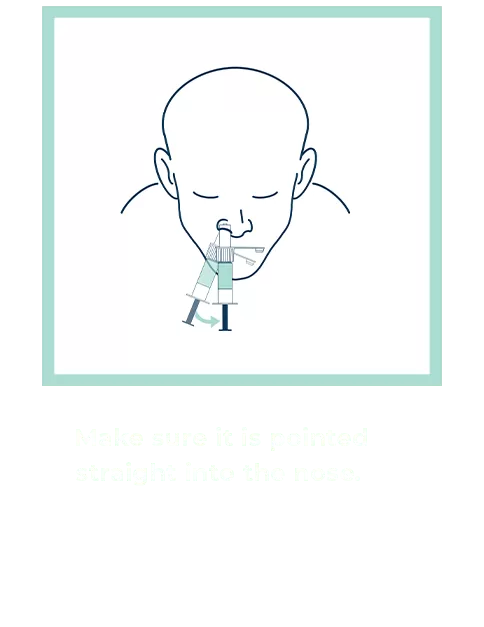

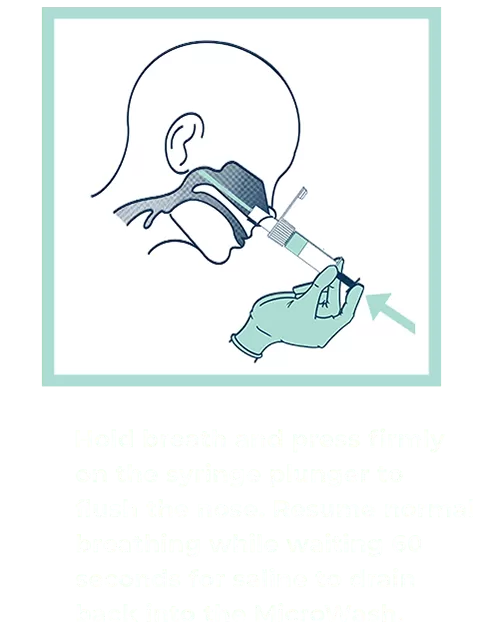

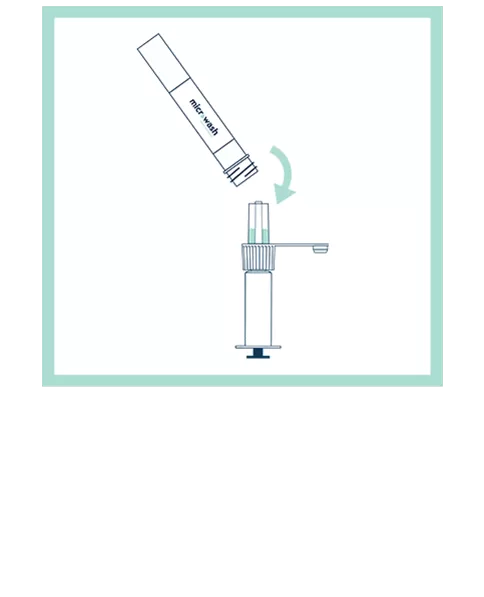

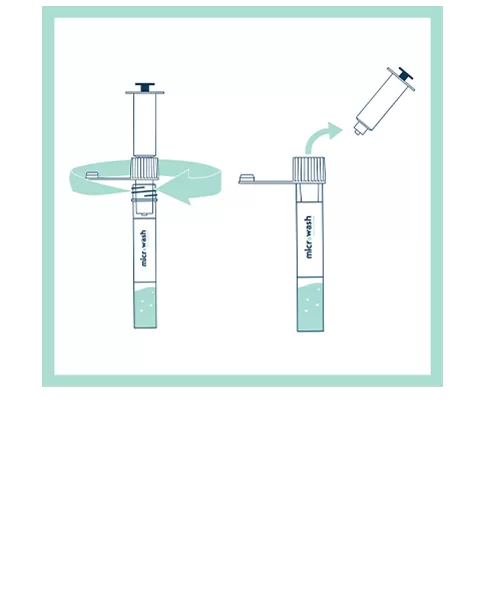
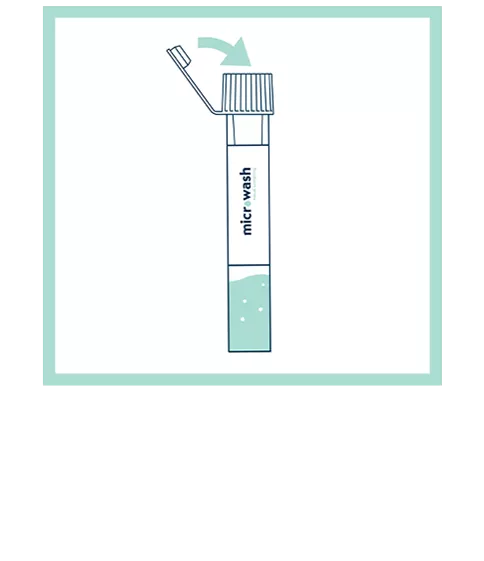




















FAQs for Collection
FAQs for Labs
More For Labs: Discover the Process & Advantage
See how our new gold standard for nasal sampling surpasses swabs and prior saline irrigation methods
on virtually all levels, and find answers to your lab processing questions.
See how our new gold standard for nasal sampling surpasses swabs and prior saline irrigation methods on virtually all levels, and find answers to your lab processing questions.

